Who came up with the plum pudding model?
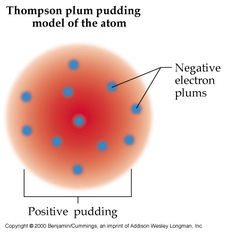
J.J. Thomson came up with the plum pudding model in the late 1800s. He envisioned that the atom consisted of a fluid that was positively charged with negatively charged electrons floating in the fluid. He is also credited with the discovery of the electron. He discovered the electrons by using the cathode ray tube. Inside the cathode ray tube, there is an electric current. Nobody at that time was quite sure what the waves were made up of. So Thomson experimented with the cathode ray tube, and electric and magnetic fields only to find that they affected the position of the rays. Therefore, he reasoned that the electric current was made of small particles, which he called corpuscles. Eventually scientists realized that the electric current was actually made up of electrons. Thomson later reasoned that the "corpuscles" must be coming form the atom itself. This showed that Dalton's solid sphere model may not actually be accurate, and that the atom isn't a solid indivisible sphere!! Pretty neat.
Hold on, what is a cathode ray rube?
A cathode ray tube is a visual display unit, used in devices such as conventional televisions (the "tube television"). It is a glass tube in which there is no air. Inside of the tube there are two metal plates, and when they are connected to a high voltage source, the plate with the negative charge, called a cathode, emits a ray. This ray is attracted to the anode, the positively charged plate right next to the cathode. The ray passes through a hole in the anode, where it travels and eventually hits a florescent screen, where it creates a bright light.
Why is it called the Plum Pudding Model?
This is because at that time in England, Plum Pudding was a popular dessert! See below
J.J. Thomson came up with the plum pudding model in the late 1800s. He envisioned that the atom consisted of a fluid that was positively charged with negatively charged electrons floating in the fluid. He is also credited with the discovery of the electron. He discovered the electrons by using the cathode ray tube. Inside the cathode ray tube, there is an electric current. Nobody at that time was quite sure what the waves were made up of. So Thomson experimented with the cathode ray tube, and electric and magnetic fields only to find that they affected the position of the rays. Therefore, he reasoned that the electric current was made of small particles, which he called corpuscles. Eventually scientists realized that the electric current was actually made up of electrons. Thomson later reasoned that the "corpuscles" must be coming form the atom itself. This showed that Dalton's solid sphere model may not actually be accurate, and that the atom isn't a solid indivisible sphere!! Pretty neat.
Hold on, what is a cathode ray rube?
A cathode ray tube is a visual display unit, used in devices such as conventional televisions (the "tube television"). It is a glass tube in which there is no air. Inside of the tube there are two metal plates, and when they are connected to a high voltage source, the plate with the negative charge, called a cathode, emits a ray. This ray is attracted to the anode, the positively charged plate right next to the cathode. The ray passes through a hole in the anode, where it travels and eventually hits a florescent screen, where it creates a bright light.
Why is it called the Plum Pudding Model?
This is because at that time in England, Plum Pudding was a popular dessert! See below
Let's move on to the Nuclear Model!